Chemlab answers5/5/2023  The reduction in the volume of the gas means that the molecules are striking the walls more often increasing the pressure, and conversely if the volume increases the distance the molecules must travel to strike the walls increases and they hit the walls less often thus decreasing the pressure. Think of it this way, if you increase the volume of a gas and must keep the pressure constant the only way to achieve this is for the temperature of the gas to increase as well.Ĭalculations using Charles' Law involve the change in either temperature (T 2) or volume (V 2) from a known starting amount of each (V 1 and T 1):īoyle's Law - states that the volume of a given amount of gas held at constant temperature varies inversely with the applied pressure when the temperature and mass are constant.  
This means that the volume of a gas is directly proportional to its Kelvin temperature. So concentrate on understanding the relationships rather than memorizing the names.Ĭharles' Law- gives the relationship between volume and temperature if the pressure and the amount of gas are held constant: 1) If the Kelvin temperature of a gas is increased, the volume of the gas increases. (P, n Constant)Ģ) If the Kelvin temperature of a gas is decreased, the volume of the gas decreases. While it is important to understand the relationships covered by each law, knowing the originator is not as important and will be rendered redundant once the combined gas law is introduced. There are 4 general laws that relate the 4 basic characteristic properties of gases to each other. 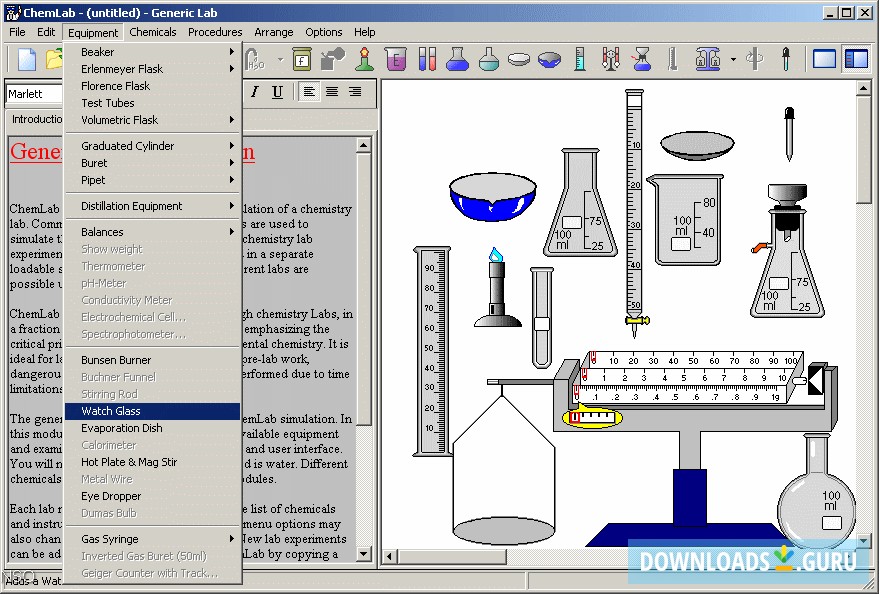
In this lecture we cover the Gas Laws: Charles',Boyle's,Avagadro's and Gay Lussacs as well as the Ideal and Combined Gas Laws. The content that follows is the substance of lecture 18. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
